Lesson 6 - Making Carbonic Acid
Lesson Overview:
Students will become familiar with the concepts of acidity, alkalinity, and pH. They will demonstrate how carbonic acid is formed and describe how it can dissolve limestone and create caves.
Objectives:
Students will be able to:
Standards Addressed:
National Science Education Standards: 5th-8th grade
Duration of Lesson/Time Requirement: 40 minutes
Materials Required:
Examples of acids and bases (if available)
Picture/Diagram of the pH scale
Can of soda pop
Safety goggles (1 per student)
For each student or pair/small group of students:
Activity #2 Part 1:
Classroom Technology:
Computer with Internet connection and projection capabilities and/or SMART Board (optional)
Safety:
This lesson should take place in a well ventilated area. Students should wear properly fitting safety goggles during Activity #2 and Activity #3 and wash their hands afterwards. Universal indicator solution is alcohol-based and flammable. Please consult universal indicator solution material safety data sheet (MSDS): http://www.sciencelab.com/msds.php?msdsId=9927029.
Read and follow all safety warnings on material labels.
At the conclusion of the lesson have students pour their used solutions in a common waste container. Dispose of all liquid waste down the drain or according to local regulations.
Set-Up:
Gather the materials necessary. Litmus paper and Universal Indicator Solution can be ordered from several online retailers including:
Sargent-Welch 800-727-4368 http://www.sargentwelch.com/store/
The Science Company 800-372-6726 http://www.sciencecompany.com/
Frey Scientific 800-225-3739 http://www.freyscientific.com/
Ward's Science 800-828-7777 https://www.wardsci.com/
For Activity #2 pour small amounts of white table vinegar and distilled water into small cups (e.g. Dixie cups). Make a solution of baking soda and distilled water and pour it into small cups. At least two days prior to the teaching of the lesson, open a can or bottle of club soda or seltzer water and let it sit out and become “flat”. Pour small amounts of fresh and flat club soda or seltzer water into small cups.
For Activity #3 transfer universal indicator solution to smaller containers for the students to share (if necessary). Pour small amounts of distilled water into clear cups.
Procedure:
Introduction
Ask the students if they know what lemons and vinegar have in common. (They both taste sour on your tongue) Explain to the students that this is because they are both acids and acids taste sour. Explain that bases are the chemical opposites of acids. They normally taste bitter and feel soapy. Another word for a base is alkali and bases are also referred to as alkaline compounds. There are strong and weak acids and bases. Explain to the students that acids and bases are some of the most important substances on Earth.
Ask the students what acids they are familiar with. What acid did they use during the last lesson during their Bubble Test? (Hydrochloric acid or white table vinegar) Discuss and explain the characteristics of most acids.
Properties of acids include:
Examples of acids include:
Ask the students if they know what the opposite of an acid is. (A base) Ask the students if they are familiar with any bases. Discuss and explain the characteristics of most bases.
Examples of bases and alkalis (strong bases):
Students will become familiar with the concepts of acidity, alkalinity, and pH. They will demonstrate how carbonic acid is formed and describe how it can dissolve limestone and create caves.
Objectives:
Students will be able to:
- Explain the concepts of acidity, alkalinity, and pH.
- Explain that carbon dioxide reacts chemically with water to form carbonic acid.
- Demonstrate how carbonic acid is formed by using the color change of universal indicator to monitor the changing pH of a solution during a chemical reaction.
- Describe how carbonic acid can dissolve limestone and create caves.
Standards Addressed:
National Science Education Standards: 5th-8th grade
- Content Standard A: Science as Inquiry
- Content Standard B: Physical Science
- Content Standard D: Earth and Space Science
Duration of Lesson/Time Requirement: 40 minutes
Materials Required:
Examples of acids and bases (if available)
Picture/Diagram of the pH scale
Can of soda pop
Safety goggles (1 per student)
For each student or pair/small group of students:
Activity #2 Part 1:
- Small cup of white table vinegar (acidic pH)
- Small cup of distilled water (neutral pH)
- Small cup of baking soda/distilled water solution (basic pH)
- 3 pieces of red litmus paper
- 3 pieces of blue litmus paper
- Dropper or pipette
- Making Carbonic Acid Student Worksheet (1 per student)
- Small cup of fresh club soda or seltzer water
- Small cup of a flat club soda or seltzer water
- 2 pieces of red litmus paper
- 2 pieces of blue litmus paper
- Small clear cup of distilled water (1 per student)
- Drinking straw (1 per student)
- Universal indicator solution
- Universal indicator pH color chart (Displayed for classroom use)
Classroom Technology:
Computer with Internet connection and projection capabilities and/or SMART Board (optional)
Safety:
This lesson should take place in a well ventilated area. Students should wear properly fitting safety goggles during Activity #2 and Activity #3 and wash their hands afterwards. Universal indicator solution is alcohol-based and flammable. Please consult universal indicator solution material safety data sheet (MSDS): http://www.sciencelab.com/msds.php?msdsId=9927029.
Read and follow all safety warnings on material labels.
At the conclusion of the lesson have students pour their used solutions in a common waste container. Dispose of all liquid waste down the drain or according to local regulations.
Set-Up:
Gather the materials necessary. Litmus paper and Universal Indicator Solution can be ordered from several online retailers including:
Sargent-Welch 800-727-4368 http://www.sargentwelch.com/store/
The Science Company 800-372-6726 http://www.sciencecompany.com/
Frey Scientific 800-225-3739 http://www.freyscientific.com/
Ward's Science 800-828-7777 https://www.wardsci.com/
For Activity #2 pour small amounts of white table vinegar and distilled water into small cups (e.g. Dixie cups). Make a solution of baking soda and distilled water and pour it into small cups. At least two days prior to the teaching of the lesson, open a can or bottle of club soda or seltzer water and let it sit out and become “flat”. Pour small amounts of fresh and flat club soda or seltzer water into small cups.
For Activity #3 transfer universal indicator solution to smaller containers for the students to share (if necessary). Pour small amounts of distilled water into clear cups.
Procedure:
Introduction
Ask the students if they know what lemons and vinegar have in common. (They both taste sour on your tongue) Explain to the students that this is because they are both acids and acids taste sour. Explain that bases are the chemical opposites of acids. They normally taste bitter and feel soapy. Another word for a base is alkali and bases are also referred to as alkaline compounds. There are strong and weak acids and bases. Explain to the students that acids and bases are some of the most important substances on Earth.
Ask the students what acids they are familiar with. What acid did they use during the last lesson during their Bubble Test? (Hydrochloric acid or white table vinegar) Discuss and explain the characteristics of most acids.
Properties of acids include:
- Corrosive (“burns” your skin)
- Sour taste (like lemons and vinegar)
- Frequently feel “sticky”
- Contains hydrogen ions when dissolved in water
- Has a pH of less than 7
- Turns blue litmus paper to a red color
- Reacts with bases to form salt and water
- Reacts with most metals to form hydrogen gas
- Reacts with carbonates to form carbon dioxide, water, and a salt
- Usually gases or liquids
Examples of acids include:
- Ascorbic acid (Vitamin C) in fruit
- Citric acid in oranges and lemons
- Acetic acid in vinegar
- Tannic acid (in tea and wine)
- Tartaric acid (in grapes)
- Hydrochloric acid (HCl) is gastric juice
- Sulfuric acid (H2SO4) is battery acid
- Nitric acid (HNO3)
- Carbonic acid (H2CO3) in soda pop
- Uric acid in urine
Ask the students if they know what the opposite of an acid is. (A base) Ask the students if they are familiar with any bases. Discuss and explain the characteristics of most bases.
- Properties of a bases include:
- Corrosive (“burns” your skin)
- Feels soapy or slippery
- Taste bitter (like baking soda)
- Has pH more than 7
- Turns red litmus paper to a blue color
- Many alkalis (soluble bases) contain hydroxyl ions (OH-)
- Reacts with oils and greases (that’s why they’re used as drain and window cleaners)
- Reacts with acids to form salt and water
- Frequently solids (although ammonia is a gas that’s usually dissolved in water)
Examples of bases and alkalis (strong bases):
- Many bleaches, soaps, toothpastes, and cleaning agents
- Magnesium hydroxide (Mg(OH)2) or milk of magnesia
- Ammonium hydroxide (NH4OH) or ammonia water
- Calcium hydroxide (Ca(OH)2) or limewater
- Sodium hydroxide (NaOH) or caustic soda/lye
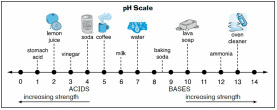
Activity #1
Display or draw the pH scale for the students to view. Ask the students if they know what it is and if they have ever heard the term “pH”. Ask the students if they can define pH. Explain to the students that scientists use something called the pH scale to measure how acidic or basic (also known as alkaline) something is. The pH scale measures acidity in the same way that a ruler measures length. pH stands for potential hydrogen and is a measurement of the hydrogen ion (H+) concentration in a liquid. If there is a high H+ concentration, the pH indicates that a solution is very acidic. If the solution is neutral, there is only a small H+ concentration, and the pH reflects that. If the solution is basic or alkaline, there is almost no H+ concentration.
Explain to the students that pH is nothing more than a way of telling how concentrated a hydrogen ion solution is. The pH scale ranges from 0 (strongly acidic) to 14 (strongly basic/alkaline). A concentration that is neither acidic nor basic is neutral and registers in the middle of the pH scale. Use the concept of money to communicate an understanding of acid/base strength to the students. Assign a neutral pH (7) the value of 1 cent. A pH of 6 (or 8) is ten times stronger, so it is worth 10 cents. A pH of 5 (or 9) is ten times stronger than that, so it is worth 100 cents (or $1). A pH of 4 (or 10) is 10 times stronger than that, so it is worth $10.
Display or draw the pH scale for the students to view. Ask the students if they know what it is and if they have ever heard the term “pH”. Ask the students if they can define pH. Explain to the students that scientists use something called the pH scale to measure how acidic or basic (also known as alkaline) something is. The pH scale measures acidity in the same way that a ruler measures length. pH stands for potential hydrogen and is a measurement of the hydrogen ion (H+) concentration in a liquid. If there is a high H+ concentration, the pH indicates that a solution is very acidic. If the solution is neutral, there is only a small H+ concentration, and the pH reflects that. If the solution is basic or alkaline, there is almost no H+ concentration.
Explain to the students that pH is nothing more than a way of telling how concentrated a hydrogen ion solution is. The pH scale ranges from 0 (strongly acidic) to 14 (strongly basic/alkaline). A concentration that is neither acidic nor basic is neutral and registers in the middle of the pH scale. Use the concept of money to communicate an understanding of acid/base strength to the students. Assign a neutral pH (7) the value of 1 cent. A pH of 6 (or 8) is ten times stronger, so it is worth 10 cents. A pH of 5 (or 9) is ten times stronger than that, so it is worth 100 cents (or $1). A pH of 4 (or 10) is 10 times stronger than that, so it is worth $10.
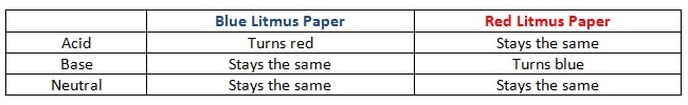
Activity #2
Part 1:
Tell the students that they are going to test some common household substances to see if they are acidic, basic, or neutral. Explain safety procedures to be followed in the classroom. Tell the students that they are to keep their safety goggles on at all times. Give each student or pair/small group of students a small cup of white table vinegar, a small cup of distilled water, a small cup of baking soda/distilled water solution, 3 pieces of red litmus paper, 3 pieces of blue litmus paper, and a dropper or pipette. Pass out a copy of the Student Worksheet to each student.
Tell the students that the small strips of red and blue paper are called litmus paper. Explain that litmus paper has been treated with a fungi that sensitizes the paper to the acidic or basic qualities of a substance. By placing a drop or two of each sample onto the litmus paper they will not know the exact pH of the solution but they will be able to get a rough estimation as to whether each substance is an acid, a base, or neutral.
Explain to the students how to use the litmus paper and what the color change of the litmus paper means.
One way the students can help remember is to think: Red = Acid; Blue = Base.
Tell the students that they are to use the dropper to place one or two drops of each solution on a strip of red litmus paper and on a strip of blue litmus paper. They will then observe and record their results on their copy of the Making Carbonic Acid Student Worksheet. Explain to the students that the color change should be instantaneous but caution them to look at the results carefully. Discuss the results and review some of the common acids and bases and their associated pH. (white table vinegar is an acid; distilled water is neutral; baking soda is a base)
Part 1:
Tell the students that they are going to test some common household substances to see if they are acidic, basic, or neutral. Explain safety procedures to be followed in the classroom. Tell the students that they are to keep their safety goggles on at all times. Give each student or pair/small group of students a small cup of white table vinegar, a small cup of distilled water, a small cup of baking soda/distilled water solution, 3 pieces of red litmus paper, 3 pieces of blue litmus paper, and a dropper or pipette. Pass out a copy of the Student Worksheet to each student.
Tell the students that the small strips of red and blue paper are called litmus paper. Explain that litmus paper has been treated with a fungi that sensitizes the paper to the acidic or basic qualities of a substance. By placing a drop or two of each sample onto the litmus paper they will not know the exact pH of the solution but they will be able to get a rough estimation as to whether each substance is an acid, a base, or neutral.
Explain to the students how to use the litmus paper and what the color change of the litmus paper means.
One way the students can help remember is to think: Red = Acid; Blue = Base.
Tell the students that they are to use the dropper to place one or two drops of each solution on a strip of red litmus paper and on a strip of blue litmus paper. They will then observe and record their results on their copy of the Making Carbonic Acid Student Worksheet. Explain to the students that the color change should be instantaneous but caution them to look at the results carefully. Discuss the results and review some of the common acids and bases and their associated pH. (white table vinegar is an acid; distilled water is neutral; baking soda is a base)
Part 2:
Hold up a can of soda pop and have the students raise their hands if they have ever had soda pop before. Ask the students if they think soda pop is acidic, basic, or neutral. Why? Explain to the students that they are going to test some club soda with litmus paper to find out if it is acidic, basic, or neutral. Tell them that club soda is just like the soda pop that they drink but it does not have any flavoring in it. Give each student or small group of students a small cup of club soda, one piece of red litmus paper, and one piece of blue litmus paper. Have the students test the club soda with their litmus paper by allowing them to dip the end of the piece of litmus paper into the club soda. Ask the students to record their observations on their Making Carbonic Acid Student Worksheets.
Once all of the students or small groups of students have tested their club soda ask them what color the litmus paper turned. Why? (Club soda is slightly acidic due to carbonation) Ask the students if they know why soda pop is fizzy. Explain to the students that soda pop is mostly carbonated water. Carbonated water is water that is mixed with carbon dioxide under pressure.
Ask the students what the chemical formula is for water and write H2O on the board. Explain the chemical formula by telling the students that water is made up of one oxygen and two hydrogen atoms. Ask the students what chemical transformation needs to take place in order for the water to become acidic. (The hydrogen needs to separate from the oxygen) Have the students take a deep breath in and then exhale. Explain to the students that when we exhale our bodies release carbon dioxide (CO2). Ask the students what happens when water mixes with carbon dioxide. Write the equation on the board: H2O + CO2 = H2CO3 (carbonic acid). Explain that the hydrogen ions are now bonded to carbon instead of oxygen. The water is now acidic or a weak carbonic acid. Tell the students that carbonic acid is what makes soda pop fizzy.
Ask the students what happens when soda pop is left out on the counter. (It loses carbon dioxide and goes flat) Give each student or small group of students a small cup of flat club soda, one piece of red litmus paper, and one piece of blue litmus paper. Have the students test the flat soda pop with the litmus paper and record their observations on their Student Worksheets. Ask the students what color the litmus paper turned. Why? Discuss the results with the students. (Flat club soda is neutral due to lack of carbonation)
Hold up a can of soda pop and have the students raise their hands if they have ever had soda pop before. Ask the students if they think soda pop is acidic, basic, or neutral. Why? Explain to the students that they are going to test some club soda with litmus paper to find out if it is acidic, basic, or neutral. Tell them that club soda is just like the soda pop that they drink but it does not have any flavoring in it. Give each student or small group of students a small cup of club soda, one piece of red litmus paper, and one piece of blue litmus paper. Have the students test the club soda with their litmus paper by allowing them to dip the end of the piece of litmus paper into the club soda. Ask the students to record their observations on their Making Carbonic Acid Student Worksheets.
Once all of the students or small groups of students have tested their club soda ask them what color the litmus paper turned. Why? (Club soda is slightly acidic due to carbonation) Ask the students if they know why soda pop is fizzy. Explain to the students that soda pop is mostly carbonated water. Carbonated water is water that is mixed with carbon dioxide under pressure.
Ask the students what the chemical formula is for water and write H2O on the board. Explain the chemical formula by telling the students that water is made up of one oxygen and two hydrogen atoms. Ask the students what chemical transformation needs to take place in order for the water to become acidic. (The hydrogen needs to separate from the oxygen) Have the students take a deep breath in and then exhale. Explain to the students that when we exhale our bodies release carbon dioxide (CO2). Ask the students what happens when water mixes with carbon dioxide. Write the equation on the board: H2O + CO2 = H2CO3 (carbonic acid). Explain that the hydrogen ions are now bonded to carbon instead of oxygen. The water is now acidic or a weak carbonic acid. Tell the students that carbonic acid is what makes soda pop fizzy.
Ask the students what happens when soda pop is left out on the counter. (It loses carbon dioxide and goes flat) Give each student or small group of students a small cup of flat club soda, one piece of red litmus paper, and one piece of blue litmus paper. Have the students test the flat soda pop with the litmus paper and record their observations on their Student Worksheets. Ask the students what color the litmus paper turned. Why? Discuss the results with the students. (Flat club soda is neutral due to lack of carbonation)
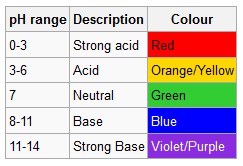
Activity #3
Tell the students that they are going to make their own carbonic acid. Explain to the students that you will be giving each of them a small clear cup filled with distilled water. Ask the students if the distilled water is an acid, a base, or neutral. (Neutral; this should be review from Activity #2 Part 1). Tell the students that for this activity they will be using a universal indicator solution instead of litmus paper. Explain to the students that a universal indicator is a solution which undergoes several color changes over a wide range of pH values. The color is used to “indicate” pH directly. Display the Universal pH Color Chart for the students to view. Ask the students what color would result in a neutral solution? (Green) An acid? (Yellow-red) A base? (Green-blue). What color should the universal indicator solution change in the distilled water? (Green)
Tell the students that once they receive their small cup of distilled water they are to add several drops of universal indicator solution and stir it into the water until the water turns noticeably green. Caution the students that universal indicator solution is alcohol-based and flammable. Remind them of safety procedures and instruct them to wear their safety goggles at all times.
Ask the students how they can make the distilled water go from being neutral to being an acid. (Add carbon dioxide) Reference the chemical equation from Activity #2 Part 2 and explain that a chemical change is required. Where can the students get carbon dioxide from? Have the students take a deep breath and exhale. (They can add carbon dioxide to the water by blowing air into it) Explain to the students that they will each received a straw. They can use the straw to stir the universal indicator solution into the distilled water. Tell the students that when they are given the signal they are to use the straw to gently blow air into the cup of distilled water. Remind the students that universal indicator solution is alcohol-based and flammable. Instruct the students that blowing too hard into the straw could result in spilling or getting the solution on them. Warn them not to inhale through the straw. This could lead to ingestion of the universal indicator solution which could cause them to get ill and have to seek medical attention.
Ask the students what they think will happen to the distilled water as they blow carbon dioxide into it. How will they know if there has been a chemical change? What does it mean if the color of the water changes? Tell the students to record their predictions on their Student Worksheet.
Pass out the materials to the students. Allow them time to mix universal indicator solution into their cups of distilled water and to record their predictions. Give the signal and allow the students to blow air into their cups. Within several seconds the students should observe a color change from green to red/orange indicating that the distilled water has become acidic. Tell the students to consult the Universal pH Color Chart if necessary and to record their observations on their Student Worksheet. Discuss the results as a class.
Note: If the educator does not feel comfortable allowing the students to complete this activity themselves it can be done as a demonstration instead.
Wrap-Up/Conclusion
Upon completion of the experiment and clean up of the students’ work stations ask the students how water might turn to carbonic acid in nature. Explain to the students that after it rains or snow melts water will travel through the soil and pick up carbon dioxide from decaying plants and other organic material. This process creates a weak carbonic acid similar to what the students just created in class. Ask the students where the carbonic acid will go once it passes through the soil. Explain that the weak carbonic acid will encounter the bedrock. Ask the students to think about what would happen if the bedrock is limestone. Explain to the students that if the bedrock is limestone (or another carbonate rock), the carbonic acid will slowly dissolve it. Ask the students why carbonic acid will dissolve limestone. (The main mineral found in limestone is calcite which dissolves in most acids) What do they think will happen if this process continues for a long period of time? (This is the process that leads to the formation of solution caves)
Tell the students that they are going to make their own carbonic acid. Explain to the students that you will be giving each of them a small clear cup filled with distilled water. Ask the students if the distilled water is an acid, a base, or neutral. (Neutral; this should be review from Activity #2 Part 1). Tell the students that for this activity they will be using a universal indicator solution instead of litmus paper. Explain to the students that a universal indicator is a solution which undergoes several color changes over a wide range of pH values. The color is used to “indicate” pH directly. Display the Universal pH Color Chart for the students to view. Ask the students what color would result in a neutral solution? (Green) An acid? (Yellow-red) A base? (Green-blue). What color should the universal indicator solution change in the distilled water? (Green)
Tell the students that once they receive their small cup of distilled water they are to add several drops of universal indicator solution and stir it into the water until the water turns noticeably green. Caution the students that universal indicator solution is alcohol-based and flammable. Remind them of safety procedures and instruct them to wear their safety goggles at all times.
Ask the students how they can make the distilled water go from being neutral to being an acid. (Add carbon dioxide) Reference the chemical equation from Activity #2 Part 2 and explain that a chemical change is required. Where can the students get carbon dioxide from? Have the students take a deep breath and exhale. (They can add carbon dioxide to the water by blowing air into it) Explain to the students that they will each received a straw. They can use the straw to stir the universal indicator solution into the distilled water. Tell the students that when they are given the signal they are to use the straw to gently blow air into the cup of distilled water. Remind the students that universal indicator solution is alcohol-based and flammable. Instruct the students that blowing too hard into the straw could result in spilling or getting the solution on them. Warn them not to inhale through the straw. This could lead to ingestion of the universal indicator solution which could cause them to get ill and have to seek medical attention.
Ask the students what they think will happen to the distilled water as they blow carbon dioxide into it. How will they know if there has been a chemical change? What does it mean if the color of the water changes? Tell the students to record their predictions on their Student Worksheet.
Pass out the materials to the students. Allow them time to mix universal indicator solution into their cups of distilled water and to record their predictions. Give the signal and allow the students to blow air into their cups. Within several seconds the students should observe a color change from green to red/orange indicating that the distilled water has become acidic. Tell the students to consult the Universal pH Color Chart if necessary and to record their observations on their Student Worksheet. Discuss the results as a class.
Note: If the educator does not feel comfortable allowing the students to complete this activity themselves it can be done as a demonstration instead.
Wrap-Up/Conclusion
Upon completion of the experiment and clean up of the students’ work stations ask the students how water might turn to carbonic acid in nature. Explain to the students that after it rains or snow melts water will travel through the soil and pick up carbon dioxide from decaying plants and other organic material. This process creates a weak carbonic acid similar to what the students just created in class. Ask the students where the carbonic acid will go once it passes through the soil. Explain that the weak carbonic acid will encounter the bedrock. Ask the students to think about what would happen if the bedrock is limestone. Explain to the students that if the bedrock is limestone (or another carbonate rock), the carbonic acid will slowly dissolve it. Ask the students why carbonic acid will dissolve limestone. (The main mineral found in limestone is calcite which dissolves in most acids) What do they think will happen if this process continues for a long period of time? (This is the process that leads to the formation of solution caves)
| lesson_6_making_carbonic_acid.pdf | |
| File Size: | 558 kb |
| File Type: | |
| lesson_6_making_carbonic_acid_student_worksheet.pdf | |
| File Size: | 253 kb |
| File Type: | |
